Microbial Control In Oilfield & Fuel Applications
Published by Leah Jones,
Digital Administrator
Oilfield Technology,
Oil producers must frequently deal with various types of microorganisms during specific processes of oil production. Aerobic and anaerobic bacteria can be present in all water-containing fluids. Studies have reported varied resistance levels to biocides commonly used in oil and gas, referring to consortia enriched from produced water samples, indicating a specific response to each biocide. This translates into the need for various biocide chemistries or for applying the concept of combined treatments (synergistic blends). With an availability of more biocides in the ‘toolbox’, Vink Chemicals can help to bypass resistance. Vink Chemicals performed a field trial with MBO (3,3′-Methylenbis(5-methyl-1,3-oxazolidin) CAS 66204-44-2) in a matured well which successfully demonstrated the application benefits of this chemistry. The biocide was injected into the near wellbore area and allowed to soak for several hours. Tracking the produced fluids from this well in terms of residual active (specially designed field methods) versus ATP levels demonstrated the suitability of this biocide under typical field conditions.
Bacteria are present as planktonic or sessile species, and their life cycle eventually may lead to the formation of complex biofilms on appropriate surfaces. Biofilms contain a variety of different species (biocenosis). Sometimes, bacteria colonise the pores within the oil reservoir as ‘native inhabitants’. In other cases, they are introduced from the environment during oilfield operations like drilling, completion, or hydraulic fracturing (inoculation, contamination etc.). Many strains grow on oilfield equipment, for example on pumps, valves, pipelines, injection wells or tank interiors if conditions are favourable. Bacteria metabolise nutrients in the fluid and thus gain energy for living. However, their chemical reactions sometimes lead to the generation of hazardous by-products like organic acids or H2S. Usually, bacteria attack the metal of process equipment in an indirect mode, resulting in corrosion and subsequent loss of structural integrity. Sulfate reducing bacteria (SRB) metabolise sulfate sources by forming biogenic H2S leading to the complex phenomenon of ‘reservoir souring’. ‘Microbiologically Influenced Corrosion (MIC)’ and related biofouling causing flow restrictions may be the consequence.
To avoid high maintenance costs, production losses, off-spec export crude or even a shut-in of wells, a biocide treatment may be considered following ‘best industry practice’. Biocides can help to mitigate and control bio growth in subsurface, downhole or topside operations if the right biocide with the appropriate combination of physical and chemical properties is selected. Oilfield biocide selection may depend on solubility/partition in oil versus water phase, pH, salinity, thermal stability, efficacy against specific strains like SRBs, chemical compatibility with other applied oilfield chemicals, the formation chemistry, speed of kill, regulatory requirements, downstream processes/disposal and many other factors. Designing a tailor-made biocide treatment that matches ‘best industry practices’ requires an advanced process understanding gained by conducting a sound site survey followed by an application assessment.
A biocide treatment can be preventative or curative. In a preventative application, a biocide is applied to a moderate to low bio contaminated stream in relatively low concentrations to keep the bacteria level under control and avoid the formation of established biofilm. Water reinjected into the reservoir needs to be treated to prevent potential reservoir souring from worsening. In a curative treatment, a fouled system needs to be sanitised by a combination of mechanical cleaning/chemicals assisted biofilm removal and a relatively high concentration of biocide typically applied in a shock treatment.
In ‘reservoir souring mitigation’ or ‘well shut-in preparation’, the biocide does not only control the initial germ count in the fluid itself, but also provides a long-term residual activity to protect the reservoir over weeks/months against potential microbial recontamination and regrowth.
Biocides may be categorised as ‘quick killing’, for example, topside produced biocides and injection water treatment. The purpose is to pre-treat and decontaminate process water. A ‘best industry practice’ to reduce the initial germ load is to apply an oxidising biocide which will reduce bacteria counts within minutes (sanitation). Other treatment programmes often include organic ‘quick killing’ biocides like THPS, TTPC, Quats, DBNPA and Glut. Depending on the available contact time, other biocides like formaldehyde releasers may also be a good choice.
‘Long term’ biocidal performance ensures the protection of treated water, e.g., during storage or drilling, fracking, water flooding, EOR operations, and well shut-in, and can prevent microbial spoilage, reservoir souring, and equipment failure caused by corrosion. For this application, biocides must be compatible with challenging conditions for an extended period of time. Required protection times are from weeks up to several months. Well-known long-term preservatives are DMO and MBO.
Biocide properties must match the specific application conditions. Some biocides are not compatible with H2S present in the fluid and quickly deactivate. Others show unacceptable foaming or may act as a H2S scavenger. Charged biocides (i.e. Quats, THPS) may get physically adsorbed onto surfaces (equipment, formation) and thus may not be available for biocidal performance in the bulk phase. Others are not compatible with high salinity brines or become thermally degraded under higher process temperatures. Many biocide residuals are difficult to monitor under field conditions. Having simple and quick field test kits available can assist in tracking residuals and, in combination with established culture based microbial tests, help in optimising the dosing regime and thus improve cost effectiveness.
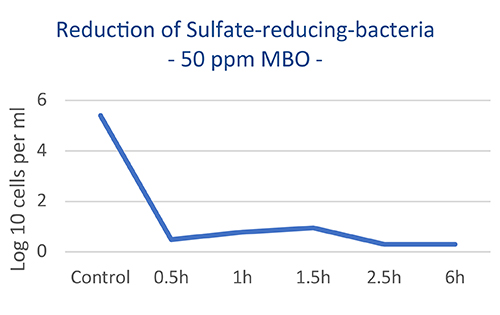
Figure 1. The experiment in Figure 1 presents biocidal efficacy against planktonic SRBs, using a mixed bacterial consortium from a production in the North Sea. The culture was grown at 30°C in a 50 000 mg/L TDS brine. The numbers were assayed using triplicate ‘most probable number’ counts. The data reveals an excellent performance at only 50 ppm dosage of MBO shown by a significant SRB reduction after 30 minutes of contact time.
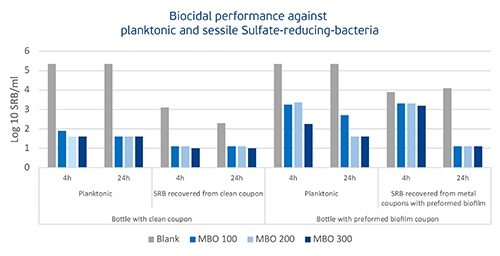
Figure 2. Figure 2 represents the biocidal efficacy of MBO against planktonic and sessile SRB. For this experiment, two bottles were equipped with (A) a clean metal coupon (to check performance against planktonic SRB/biofilm formation inhibition capability) and (B) a coupon with preformed SRB biofilm (to check efficacy against planktonic SRB and detachment/killing of SRB biofilm). The recovered SRB were enumerated by serial dilution (MPN method; NACE TM0-194 (2014)). The graph shows MBO’s excellent biocidal performance against planktonic SRB, inhibiting the growth of an SRB biofilm, and detaching and killing preformed SRB biofilm. To eliminate planktonic SRB within four hours of contact time, a dosage of 100 ppm MBO was sufficient. Performance against SRB Biofilm requires either 100 ppm/24 hours or 300 ppm/4 hours of MBO.
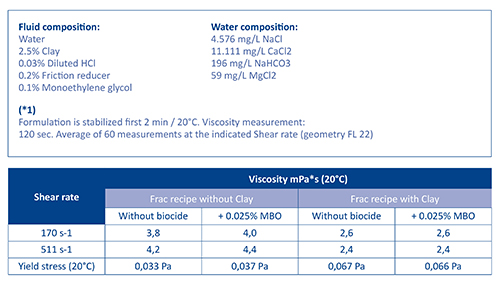
Figure 3. Figure 3 shows that a 0.025% MBO dosage has no negative impact on viscosity properties under the test conditions with this specific frac formulation. Additional retesting after three days of storage confirmed the initial results.

Figure 4. Figure 4 presents the stability and long-term activity of MBO compared to the commonly used biocide glutaraldehyde. Both biocides were mixed with a frac water and stored over a period of 70 days at elevated temperatures of 75°C. Long-term stability was proven after the storage period by checking the biocidal activity. The graph shows the excellent long-term activity of MBO in frac water.

Figure 5.The goal of the test in Figure 5 was to identify a biocide that could effectively reduce the initial germ load, handle a rechallenge, and eventually offer full protection of the system by inhibition of regrowth of bacteria over the entire duration of the test. As seen in the graph, a dosage of 250 ppm DMO (a. i.) shows only limited germ reduction after the first and second inoculation. Obviously, this dosage is not sufficient to inhibit regrowth of bacteria, so after a couple of weeks, the system is fully contaminated. In contrast, 250 ppm (a. i.) MBO shows full biocidal efficacy after 24 hours of contact time after the first inoculation. The same performance is detected after the reinoculation. Additionally, this dosage is sufficient to protect the system against regrowth of bacteria over a period of approximately two months. To achieve all goals outlined in this test, either 500 ppm (a. i.) of the biocide DMO or 250 ppm (a. i.) of MBO is required. Since DMO is commercially available as a 74% aqueous solution, this will correspond to a higher dosage rate of 676 ppm.

Figure 6. Field test data/North Africa Region.
To find out more information on Vink Chemicals Oilfield & Fuel additives please watch here or visit their website: www.vink-chemicals.com.
Authors:
Jennifer Knopf, Product Development Manager Oilfield & Fuel, Vink Chemicals Dr. Ulf W. Naatz, Head of Technical Applications Oilfield & Fuel, Vink Chemicals.
Read the article online at: https://www.oilfieldtechnology.com/special-reports/01062022/microbial-control-in-oilfield-fuel-applications/
You might also like
EnerMech wins major subsea pre-commissioning contract for Subsea7 on Trion development
This latest award reinforces EnerMech’s strong capability in the Americas and its commitment to supporting clients with technical expertise, operational reliability and integrated solutions that improve certainty across complex offshore developments.


